Chloroacetone, reasonably free from higher chlorinated products, was made by chlorinating 2320 g. (2900 ml.; 40 moles) of c. p. acetone with a relatively small amount (320 g. = 2.37 moles) of sulfuryl chloride (practical grade). The latter was added slowly with stirring from a dropping funnel over a period of five and one-half hours, taking precautions to exclude moisture and maintaining the reaction flask immersed in an ice-bath. After removal of excess acetone in vacuo, the residue was washed with aqueous potassium carbonate solution, dried over sodium sulfate (chloroacetone apparently forms a constant boiling mixture with water and acetone) and fractionated.
The yield was 158 g., 72% based on sulfuryl chloride, b. p. 118—120°, d(15/4) 1.135, d(25/4) 1.123.
A dried ether solution (approximately 500 ml.) containing 0.5 mole of diazomethane was placed in a 1000-ml. three-necked flask and Eastman Kodak practical grade acetyl chloride (0.25 mole) was added slowly from a dropping funnel with constant stirring of the solution which was maintained at a temperature not greater than 5°. The reaction mixture was allowed to stand for two hours after the addition of the acetyl chloride and was then saturated with dry hydrogen chloride over a period of two hours. The bulk of the ether was removed by distillation and the residual solution fractionated through a small column. The product boiling at 118—119° at 736 mm., d(25/4) 1.1260, weighed 15.8 g. (68% based on acetyl chloride).
Anal. Calcd. for C3H5OCl: Cl, 38.3. Found: Cl (Stepanov), 38.4.
A solution of 150 ml. of acetone, 50 ml. of water, 12 g. of CuCl2*2H2O and 6 g. of LiCl was refluxed for 24 hours and then rapidly distilled. Material boiling between 56 and 100° was collected and resubjected to careful distillation through a five-foot helix-packed vacuum jacketed column to remove the acetone. The lachrymatory residue (about 15 ml.) separated into two layers on cooling. The material was centrifuged and the oily lower layer was distilled through an eighteen-inch vacuum jacketed spiral column. The material was thus separated into two constant boiling fractions: I (b.p. 89°), which separated into two layers on cooling and consisted of chloroacetone and water in the ratio of approximately 5:1, and II (b.p. 118°) which is pure chloroacetone. The 2,4-dinitrophenylhydrazones of chloroacetone made in this manner did not depress the melting point (125—126°) of an authentic sample. The approximate chloroacetone yield is 60% (3.3 g).

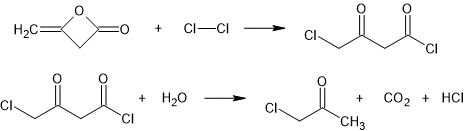
Dry chlorine gas was passed at atmospheric pressure into a body of 25 grams (0.297 moles) of diketene, maintained at a temperature of from 0 to 10 C., until 21 grams (0.296 moles) of chlorine had been absorbed. The chlorinated product then was poured into 150 c. c. of cold water, and the reaction mixture was heated under reflux for two hours, and then was allowed to cool to room temperature overnight. Carbon dioxide was evolved; and monochloroacetone separated from the solution. The reaction mixture was extracted three times with ether, the ether extract was dried over calcium chloride, and the ether was distilled from the mixture. The residue was fractionally distilled under atmospheric pressure, yielding 19.1 grams of monochloroacetone, boiling at from 117 to 121 C. under the said pressure, corresponding to a yield of about 70%.
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru